Analysis of Viral Vector, Non-Viral Vector and Gene Therapy Manufacturing market Strategies and Forecasts to 2035
Roots Analysis has announced the addition of “Viral Vector, Non-Viral Vector and Gene Therapy Manufacturing Market (5th Edition), 2022-2035” report to its list of offerings.
Key Market Insights
- Presently, over 235 industry and non-industry players claim to have the necessary capabilities to manufacture various types of viral and non-viral vectors, which can be used across different application areas
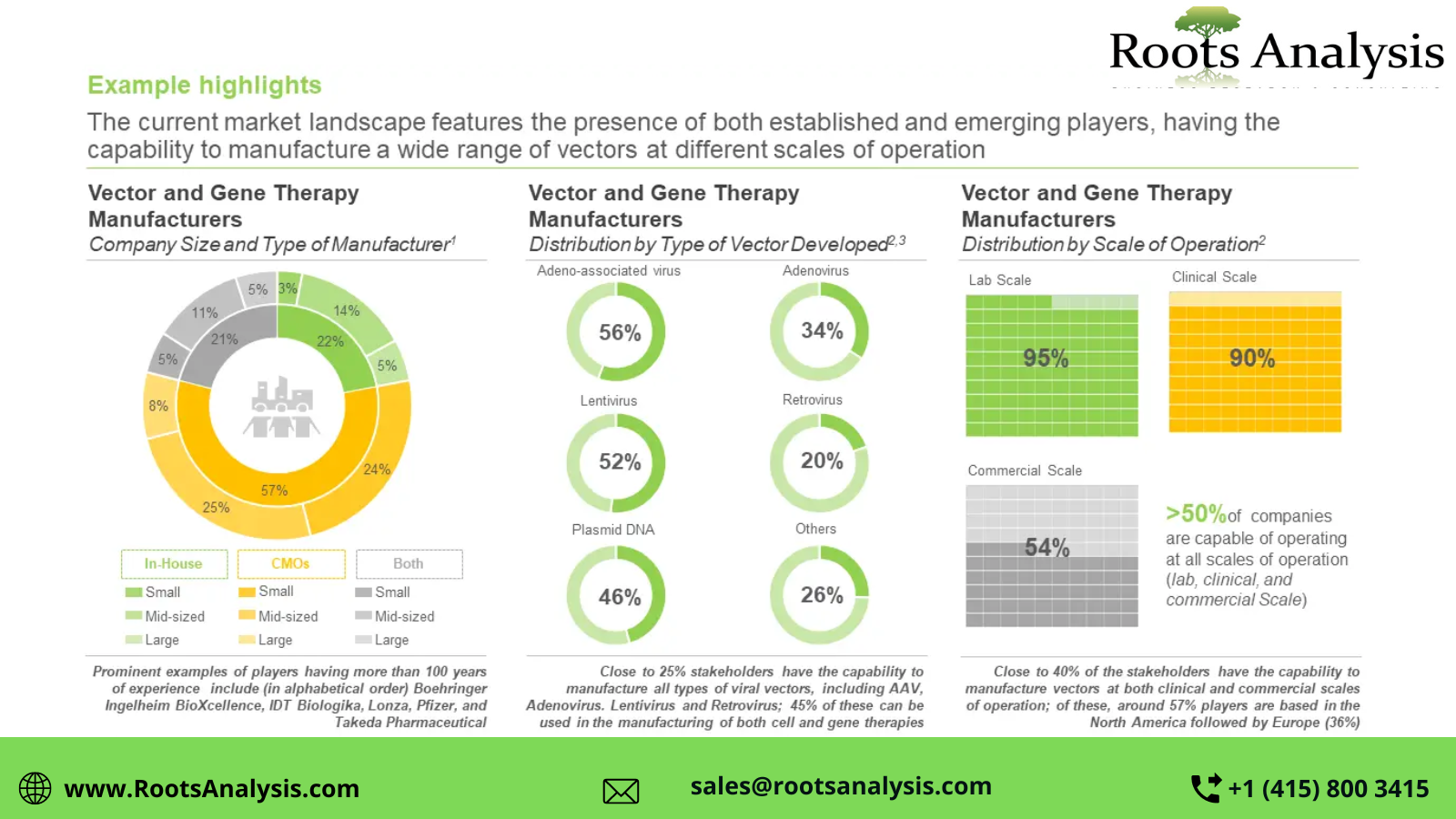
- The current market landscape features the presence of both established and emerging players, having the capability to manufacture a wide range of vectors at different scales of operation
- The supply chain, across various vector types, is well distributed across different regions; both industry and non-industry players have contributed significantly to the industry’s evolution
- Players have established their presence across various geographical regions to meet the growing demand of vectors and acquire the required capabilities; the US and Europe are the key manufacturing hubs
- In pursuit of gaining a competitive edge, manufacturers claim to be steadily expanding their existing capabilities to enhance their expertise in the vector and gene therapy related domain / service portfolios
- Over the past few years, the field has witnessed significant efforts in terms of expansions and collaborations for increasing vector manufacturing capabilities; this reflects the rising interest of stakeholders in this domain
- In order to address the growing complexities associated with the development and manufacturing of vectors, companies are actively developing / improving affiliated technological platforms
- Owing to the need for optimization of the manufacturing processes, several vector and gene therapy innovators are anticipated to forge strategic alliances with the vector and gene therapy manufactures
- Case Study: Novel viral and bacterial strains, such as Chimeric virus and Bifidobacterium longum, are currently being investigated as vectors that can be further used in various gene therapies and vaccines
- The pricing of vectors vary considerably; suppliers offer these vectors across a multitude of concentrations and different volume ranges
- Increasing clinical research and demand for the vectors, especially for therapies focused on rare and complex disease indications, is anticipated to create lucrative business opportunities
- Stakeholders engaged in the viral vector manufacturing and non-viral vectors manufacturing, presently hold a global manufacturing installed capacity of more than 200 thousand liters
- The demand for vectors is anticipated to increase at a significate pace in future and stakeholders are actively investing in establishing new facilities in order to meet the growing demand
- With the ongoing expansion initiatives, the market is expected to have sufficient capacity to meet the rising demand for vectors in the coming years
- The overall opportunity is anticipated to grow at the CAGR of 15%; it is likely to be well distributed across different scales of operation and type of vectors
- The projected opportunity for vector and gene therapy manufacturing is expected to be segmented across different therapeutic areas, application areas and key geographical regions
Table of Content
- PREFACE
1.1. Scope of the Report
1.2. Research Methodology
1.3. Key Questions Answered
1.4. Chapter Outlines
- EXECUTIVE SUMMARY
- INTRODUCTION
3.1. Chapter Overview
3.2. Viral and Non-Viral Gene Transfer Techniques
3.3. Viral Vectors Used in Genetically Modified Therapies
3.4. Types of Viral Vectors
3.4.1. Adeno-associated Viral Vectors
3.4.2. Adenoviral Vectors
3.4.3. Lentiviral Vectors
3.4.4. Retroviral Vectors
3.4.5. Other Viral Vectors
3.4.5.1. Alphavirus
3.4.5.2. Foamy Virus
3.4.5.3. Herpes Simplex Virus
3.4.5.4. Sendai Virus
3.4.5.5. Simian Virus
3.4.5.6. Vaccinia Virus
3.5. Types of Non-Viral Vectors
3.5.1. Plasmid DNA
3.5.2. Liposomes, Lipoplexes and Polyplexes
3.5.3. Oligonucleotides
3.5.4. Other Non-Viral Vectors
3.6. Gene Delivery using Non-Viral Vectors
3.6.1. Biolistic Methods
3.6.2. Electroporation
3.6.3. Receptor Mediated Gene Delivery
3.6.4. Gene Activated Matrix (GAM)
3.7. Applications of Viral and Non-Viral Vectors
3.7.1. Type of Therapy
3.7.1.1. Cell and Gene Therapy
3.7.1.2. Vaccinology
3.8. Current / Ongoing Trends in Vector Development / Manufacturing
3.8.1. Vector Engineering
3.8.2. Cargo Engineering
3.9. Vector Manufacturing
3.9.1. Types of Vector Manufacturers
3.9.2. Viral Vector Manufacturing Processes
3.9.2.1 Vector Production
3.9.2.2. Adherent and Suspension Cultures
3.9.2.3. Unit Process Versus Multiple Parallel Processes
3.9.2.4. Cell Culture Systems for Production of Viral Vectors
3.9.2.5. Serum-Containing versus Serum-Free Media
3.9.3. Bioprocessing of Viral Vectors
3.9.3.1. AAV Vector Production
3.9.3.2. Adenoviral Vector Production
3.9.3.3. Lentiviral Vector Production
3.9.3.4. γ -Retroviral Vector Production
3.9.4. Key Challenges Associated with Vector Manufacturing
3.10. Future Perspectives
- VIRAL VECTOR AND GENE THERAPY MANUFACTURERS (INDUSTRY PLAYERS): MARKET LANDSCAPE
4.1. Chapter Overview
4.2. Viral Vector and Gene Therapy Manufacturers: Overall Market Landscape
4.2.1. Analysis by Year of Establishment
4.2.2. Analysis by Company Size
4.2.3. Analysis by Location of Headquarters
4.2.4. Analysis by Type of Product Manufactured
4.2.5. Analysis by Location of Vector Manufacturing Facilities
4.2.6. Analysis by Type of Manufacturer
4.2.7. Analysis by Scale of Operation
4.2.8. Analysis by Location of Headquarters and Scale of Operation
4.2.9. Analysis by Type of Vector Manufactured
4.2.10. Analysis by Scale of Operation and Type of Vector Manufactured
4.2.11. Analysis by Application Area
4.2.12. Information on Production Capacity
- PLASMID DNA AND GENE THERAPY MANUFACTURERS (INDUSTRY PLAYERS): MARKET LANDSCAPE
5.1. Chapter Overview
5.2. Plasmid DNA and Gene Therapy Manufacturers: Overall Market Landscape
5.2.1. Analysis by Year of Establishment
5.2.2. Analysis by Company Size
5.2.3. Analysis by Location of Headquarters
5.2.4. Heat Map: Analysis by Company Size and Location of Headquarters
5.2.5. Analysis by Type of Product Manufactured
5.2.6. Analysis by Location of Plasmid DNA Manufacturing Facilities
5.2.7. Analysis by Type of Manufacturer
5.2.8. Analysis by Scale of Operation
5.2.9. Analysis by Application Area
5.2.10. Information on Production Capacity
- VECTOR AND GENE THERAPY MANUFACTURERS (NON-INDUSTRY PLAYERS): MARKET LANDSCAPE
6.1. Chapter Overview
6.2. Vector and Gene Therapy Manufacturers: Overall Market Landscape
6.2.1. Analysis by Year of Establishment
6.2.2. Analysis by Location of Vector Manufacturing Facilities
6.2.3. Analysis by Type of Manufacturer
6.2.4. Analysis by Scale of Operation
6.2.5. Analysis by Type of Vector Manufactured
6.2.6. Analysis by Scale of Operation and Type of Vector Manufactured
6.2.7. Analysis by Application Area
- VECTOR AND GENE THERAPY MANUFACTURING TECHNOLOGIES: MARKET LANDSCAPE
7.1. Chapter Overview
7.2. Vector and Gene Therapy Manufacturing Technologies
7.2.1. Analysis by Type of Technology
7.2.2. Analysis by Purpose of Technology
7.2.3. Analysis by Scale of Operation
7.2.4. Analysis by Type of Vector
7.2.5. Analysis by Application Area
7.2.6. Most Active Players: Analysis by Number of Technology
7.3. Concluding Remarks
- COMPANY COMPETITIVENESS ANALYSIS
8.1. Chapter Overview
8.2. Methodology and Key Parameters
8.3. Vector and Gene Therapy: In-House Manufacturers
8.3.1. Players based in North America
8.3.2. Players based in Europe
8.3.3. Players based in Asia-Pacific and Rest of the World
8.4. Vector and Gene Therapy: Contract Manufacturing Organizations
8.4.1. Players based in North America
8.4.2. Players based in Europe
8.4.3. Players based in Asia-Pacific and Rest of the World
8.5. Vector and Gene Therapy: Players Engaged in In-house and Contract Manufacturing
8.5.1. Players based in North America
8.5.2. Players based in Europe
8.5.3. Players based in Asia-Pacific and Rest of the World
- VECTOR AND GENE THERAPY MANUFACTURERS IN NORTH AMERICA
9.1. Chapter Overview
9.2. Advanced BioScience Laboratories
9.2.1. Company Overview
9.2.2. Manufacturing Facilities
9.2.3. Recent Developments and Future Outlook
9.3. Aldevron
9.3.1. Company Overview
9.3.2. Financial Information
9.3.3. Manufacturing Facilities
9.3.4. Manufacturing Experience
9.3.5. Recent Developments and Future Outlook
9.4. BioReliance / SAFC Commercial (Merck KGaA)
9.4.1. Company Overview
9.4.2. Financial Information
9.4.3. Vector Manufacturing Technology Portfolio
9.4.4. Manufacturing Facilities
9.4.5. Recent Developments and Future Outlook
9.5. bluebird bio
9.5.1. Company Overview
9.5.2. Financial Information
9.5.3. Manufacturing Facilities
9.5.4. Manufacturing Experience
9.5.5. Recent Development and Future Outlook
9.6. Other Companies
9.6.1. Audentes Therapeutics
9.6.1.1. Company Overview
9.6.1.2. Financial Information
9.6.1.3. Manufacturing Facilities
9.6.1.4. Recent Developments and Future Outlook
9.6.2. Emergent BioSolutions
9.6.2.1. Company Overview
9.6.2.2. Financial Information
9.6.2.3 Manufacturing Facilities
9.6.2.4. Recent Developments and Future Outlook
9.6.3. FUJIFILM Diosynth Biotechnologies
9.6.3.1. Company Overview
9.6.3.2. Financial Information
9.6.3.3. Manufacturing Facilities
9.6.3.4. Manufacturing Experience
9.6.3.5. Recent Developments and Future Outlook
9.6.4. MeiraGTx
9.6.4.1. Company Overview
9.6.4.2. Manufacturing Facilities
9.6.4.3. Recent Developments and Future Outlook
9.6.5. Spark Therapeutics
9.6.5.1. Company Overview
9.6.5.2. Financial Information
9.6.5.3. Manufacturing Facilities
9.6.5.4. Vector Manufacturing Technology Portfolio
9.6.5.5. Manufacturing Experience
9.6.5.6. Recent Developments and Future Outlook
9.6.7. Vigene Biosciences
9.6.7.1. Company Overview
9.6.7.2. Manufacturing Facilities
9.6.7.3. Vector Manufacturing Technology Portfolio
9.6.7.4. Manufacturing Experience
9.6.7.5. Recent Developments and Future Outlook
- VECTOR AND GENE THERAPY MANUFACTURERS IN EUROPE
10.1. Chapter Overview
10.2. Biovian
10.2.1. Company Overview
10.2.2. Manufacturing Facilities
10.2.3. Recent Developments and Future Outlook
10.3. Centre for Process Innovation
10.3.1. Company Overview
10.3.2. Manufacturing Facilities
10.3.3. Recent Developments and Future Outlook
10.4. Cobra Biologics
10.4.1. Company Overview
10.4.2. Financial Information
10.4.3. Manufacturing Facilities
10.4.4. Vector Manufacturing Technology Portfolio
10.4.5. Manufacturing Experience
10.4.6. Recent Developments and Future Outlook
10.5. FinVector
10.5.1. Company Overview
10.5.2. Manufacturing Facilities
10.5.3. Vector Manufacturing Technology Portfolio
10.5.4. Manufacturing Experience
10.5.5. Recent Developments and Future Outlook
10.6. Kaneka Eurogentec
10.6.1. Company Overview
10.6.2. Manufacturing Facilities
10.6.3. Manufacturing Experience
10.6.4. Recent Developments and Future Outlook
10.7. Lonza
10.7.1. Company Overview
10.7.2. Financial Information
10.7.3. Vector Manufacturing Technology Portfolio
10.7.4. Manufacturing Facilities
10.7.5. Recent Developments and Future Outlook
10.8. MolMed
10.8.1. Company Overview
10.8.2. Financial Information
10.8.3. Manufacturing Facilities
10.8.4. Recent Developments and Future Outlook
10.9. Novasep
10.9.1. Company Overview
10.9.2. Financial Information
10.9.3. Manufacturing Facilities
10.9.4. Manufacturing Experience
10.9.5. Recent Developments and Future Outlook
10.10. Orchard Therapeutics
10.10.1. Company Overview
10.10.2. Manufacturing Facilities
10.10.3 Recent Developments and Future Outlook
10.11. Oxford BioMedica
10.11.1. Company Overview
10.11.2. Financial Information
10.11.3. Manufacturing Facilities
10.11.4. Vector Manufacturing Technology Portfolio
10.11.5. Manufacturing Experience
10.11.6. Recent Developments and Future Outlook
10.12. Richter-Helm
10.12.1. Company Overview
10.12.2. Manufacturing Facilities
10.12.3. Recent Developments and Future Outlook
10.13. Sanofi (CEPiA, Sanofi Pasteur, Genzyme)
10.13.1. Company Overview
10.13.2. Financial Information
10.13.3. Manufacturing Facilities
10.13.4. Recent Developments and Future Outlook
10.14. uniQure
10.14.1. Company Overview
10.14.2. Financial Information
10.14.3. Manufacturing Facilities
10.14.4. Vector Manufacturing Technology Portfolio
10.14.5. Recent Developments and Future Outlook
10.15. Vibalogics
10.15.1. Company Overview
10.15.2. Manufacturing Facilities
10.15.3. Recent Developments and Future Outlook
10.16. VIVEbiotech
10.16.1. Company Overview
10.16.2. Vector Manufacturing Technology Portfolio
10.16.3. Manufacturing Facilities
10.16.4. Recent Developments and Future Outlook
10.17. Other Companies
10.17.1. Cell and Gene Therapy Catapult
10.17.1.1. Company Overview
10.17.1.2. Manufacturing Facilities
10.17.1.3. Recent Developments and Future Outlook
- VECTOR AND GENE THERAPY MANUFACTURERS IN ASIA-PACIFIC
11.1. Chapter Overview
11.2. Wuxi AppTec
11.2.1. Company Overview
11.2.2. Financial Information
11.2.3. Manufacturing Facilities
11.2.4. Manufacturing Experience
11.2.5. Recent Developments and Future Outlook
11.3. Other Key Players
- OTHER KEY PLAYERS
12.1. Chapter Overview
12.2. Thermo Fisher Scientific
12.2.1. Company Overview
12.2.2. Financial Information
12.2.3. Vector Manufacturing Related Capabilities
12.2.4. Recent Developments and Future Outlook
12.3. BioNTech Innovative Manufacturing
12.3.1. Company Overview
12.3.2. Financial Information
12.3.3. Vector Manufacturing Related Capabilities
12.3.4. Recent Developments and Future Outlook
12.3. Celonic
12.3.1. Company Overview
12.3.2. Vector Manufacturing Related Capabilities
12.3.3. Recent Developments and Future Outlook
- RECENT PARTNERSHIPS
13.1. Chapter Overview
13.2. Partnership Models
13.3. Vector and Gene Therapy Manufacturing: Recent Partnerships
13.3.1. Analysis by Year of Partnership
13.3.2. Analysis by Type of Partnership
13.3.3. Analysis by Scale of Operation
13.3.4. Analysis by Type of Vector
13.3.5. Analysis by Therapeutic Area
13.3.6. Most Active Players: Analysis by Number of Partnerships
13.3.7. Geographical Analysis
13.3.7.1. Intercontinental and Intracontinental Agreements
13.4. Other Collaborations
- RECENT EXPANSIONS
14.1. Chapter Overview
14.2. Expansions Models
14.3. Vector and Gene Therapy Manufacturing: Recent Expansions
14.3.1. Analysis by Year of Expansion
14.3.2. Analysis by Type of Expansion
14.3.3. Analysis by Amount Invested by Key Players
14.3.4. Analysis by Scale of Operation
14.3.5. Analysis by Type of Vector
14.3.6. Analysis by Application Area
14.3.7. Most Active Players: Analysis by Number of Expansions
14.3.8. Geographical Analysis
14.3.8.1. Analysis by Location of Expansion
- STRATEGIC PARTNER ANALYSIS
15.1. Chapter Overview
15.2. Strategic Partner Analysis: Viral Vector based Therapy Developers
15.3. Methodology and Key Parameters
15.3.1. Opportunities for AAV Vector- based Therapy Developers
15.3.1.1. Most Likely Partners
15.3.1.2. Likely Partners
15.3.1.3. Less Likely Partners
15.3.1.4. Least Likely Partners
15.3.2. Opportunities for Adenoviral Vector based Therapy Developers
15.3.2.1. Most Likely Partners
15.3.2.2. Likely Partners
15.3.2.3. Less Likely Partners
15.3.2.4. Least Likely Partners
15.3.3. Opportunities for Lentiviral Vector based Therapy Developers
15.3.3.1. Most Likely Partners
15.3.3.2. Likely Partners
15.3.3.3. Less Likely Partners
15.3.3.4. Least Likely Partners
15.3.4. Opportunities for Retroviral Vector based Therapy Developers
15.3.4.1. Most Likely Partners
15.3.4.2. Likely Partners
15.3.4.3. Less Likely Partners
15.3.4.4. Least Likely Partners
15.3.5. Opportunities for Other Viral Vector based Therapy Developers
15.3.5.1. Most Likely Partners
15.3.5.2. Likely Partners
15.3.5.3. Less Likely Partners
15.3.5.4. Least Likely Partners
15.4. Strategic Partner Analysis: Viral Vector based Purification Product Developers
15.5. Methodology and Key Parameters
15.5.1. Opportunities for AAV based Purification Product Developers
15.5.1.1. Most Likely Partners
15.5.1.2. Likely Partners
15.5.2. Opportunities for Adenoviral Vector based Purification Product Developers
15.5.2.1. Most Likely Partners
15.5.2.2. Likely Partners
15.5.3. Opportunities for Lentiviral Vector based Purification Product Developers
15.5.3.1. Most Likely Partners
15.5.3.2. Likely Partners
15.5.4. Opportunities for Retroviral Vector based Purification Product Developers
15.5.4.1. Most Likely Partners
15.5.4.2. Likely Partners
15.5.5. Opportunities for Other Viral Vector based Purification Product Developers
15.5.5.1. Most Likely Partners
15.5.5.2. Likely Partners
- EMERGING VECTORS
16.1. Chapter Overview
16.1.1. Alphavirus based Vectors
16.1.2. Anc80 based Vectors
16.1.3. Bifidobacterium longum based Vectors
16.1.4. Cytomegalovirus based Vectors
16.1.5. Listeria monocytogenes based Vectors
16.1.6. Minicircle DNA based Vectors
16.1.7. Myxoma Virus based Vectors
16.1.8. Self-Complementary Vectors
16.1.9. Sendai Virus based Vectors
16.1.10. Sleeping Beauty Transposons
16.1.11. Vaccinia Virus and Modified Vaccinia Ankara based Vectors
16.1.12. Chimeric Viral Vectors
- KEY INSIGHTS
17.1. Chapter Overview
17.2. Vector and Gene Therapy Manufacturers: Analysis by Purpose of Manufacturing, Type of Vector Manufactured and Scale of Operation
17.3. Vector and Gene Therapy Manufacturers: Analysis by Company Size and Type of Vector Manufactured
17.4. Vector and Gene Therapy Manufacturers: Prominent Geographical Hubs by Type of Organization
17.4.1. Contract Manufacturing Organizations
17.4.2. In-House Manufacturers
17.5. Vector and Gene Therapy Manufacturers: Analysis by Location of Manufacturing Facilities and Type of Vector Manufactured
17.5.1. AAV Vector Manufacturers
17.5.2. Adenoviral Vector Manufacturers
17.5.3. Lentiviral Vector Manufacturers
17.5.4. Retroviral Vector Manufacturers
17.5.5. Plasmid DNA Manufacturers
- COST PRICE ANALYSIS
18.1. Chapter Overview
18.2. Factors Contributing to High Price of Viral Vector and Plasmid DNA based Therapies
18.3. Viral Vector and Plasmid DNA based Therapies: Pricing Models
18.3.1. Pricing Models on the Basis of Expert Opinions
18.3.2. Pricing Models on the Basis of Manufacturing Cost
18.3.2.1. Pricing Models on the Basis of Technology Used
18.3.2.2. Pricing Models on the Basis of Scale of Manufacturing
18.3.2.3. Pricing Models on the Basis of Type of Client
18.3.3. Prices of Different Types of Vectors
18.4. Concluding Remarks
- CAPACITY ANALYSIS
19.1. Chapter Overview
19.2. Methodology and Key Assumptions
19.2. Global Installed Viral Vector and Gene Therapy Manufacturing Capacity
19.3. Global Installed Viral Vector Manufacturing Capacity
19.3.1. Analysis by Company Size
19.3.2. Analysis by Scale of Operation
19.3.3. Analysis by Location of Manufacturing Facilities
19.4. Global Installed Plasmid DNA Manufacturing Capacity
19.4.1. Analysis by Company Size
19.4.2. Analysis by Scale of Operation
19.4.3. Analysis by Location of Manufacturing Facilities
19.4. Concluding Remarks
- DEMAND ANALYSIS
20.1. Chapter Overview
20.2. Assumptions and Methodology
20.3. Global Clinical Demand for Viral Vectors and Plasmid DNA, 2022-2035
20.3.1. Analysis by Type of Vector
20.3.2. Analysis by Type of Therapy
20.3.3. Analysis by Therapeutic Area
20.3.4. Analysis by Geographical Location
20.4. Global Commercial Demand for Viral Vectors and Plasmid DNA, 2022-2035
20.4.1. Analysis by Type of Vector
20.4.2. Analysis by Type of Therapy
20.4.3. Analysis by Therapeutic Area
20.4.4. Analysis by Geographical Location
20.5. Demand and Supply Analysis
20.5.1. Scenario 1
20.5.2. Scenario 2
20.5.3. Scenario 3
- MARKET SIZING AND OPPORTUNITY ANALYSIS
21.1. Chapter Overview
21.2. Scope of the Forecast
21.3. Forecast Methodology
21.4. Input Tables and Key Assumptions
21.5. Global Viral Vector and Plasmid DNA Manufacturing Market, 2022-2035
21.5.1. Viral Vector and Plasmid DNA Manufacturing Market, 2022-2035: Distribution by Type of Vector Manufactured
21.5.1.1. Viral Vector and Plasmid DNA Manufacturing Market, 2022-2035: Market Attractiveness by Purpose of Manufacturing
21.5.2. Viral Vector and Plasmid DNA Manufacturing Market, 2022-2035: Distribution by Scale of Operation
21.5.3. Viral Vector and Plasmid DNA Manufacturing Market, 2022-2035: Distribution by Type of Vector Manufactured
21.5.4. Viral Vector and Plasmid DNA Manufacturing Market, 2022-2035: Distribution by Therapeutic Area
21.5.5. Viral Vector and Plasmid DNA Manufacturing Market, 2022-2035: Distribution by Application Area
21.5.6. Viral Vector and Plasmid DNA Manufacturing Market, 2022-2035: Distribution by Key Geographical Regions
21.6. Current and Future Market Opportunity for Commercial Products
21.6.1. Viral Vector and Plasmid DNA Manufacturing Market for Commercial Products, 2022-2035: Distribution by Type of Vector Manufactured
21.6.1.1. AAV Vectors
21.6.1.1.1. AAV Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Therapeutic Area
21.6.1.1.2. AAV Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Application Area
21.6.1.1.3. AAV Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Key Geographical Regions
21.6.1.2. Adenoviral Vectors
21.6.1.2.1. Adenoviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Therapeutic Area
21.6.1.2.2. Adenoviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Application Area
21.6.1.2.3. Adenoviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Key Geographical Regions
21.6.1.3. Lentiviral Vectors
21.6.1.3.1. Lentiviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Therapeutic Area
21.6.1.3.2. Lentiviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Application Area
21.6.1.3.3. Lentiviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Key Geographical Regions
21.6.1.4. Retroviral Vectors
21.6.1.4.1. Retroviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Therapeutic Area
21.6.1.4.2. Retroviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Application Area
21.6.1.4.3. Retroviral Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Key Geographical Regions
21.6.1.5. Plasmid DNA
21.6.1.5.1. Plasmid DNA Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Therapeutic Area
21.6.1.5.2. Plasmid DNA Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Application Area
21.6.1.5.3. Plasmid DNA Vector Manufacturing Market for Commercial Products, 2022-2035: Distribution by Key Geographical Regions
21.6.2. Viral Vector and Plasmid DNA Manufacturing Market for Commercial Products, 2022-2035: Distribution by Therapeutic Area
21.6.3. Viral Vector and Plasmid DNA Manufacturing Market for Commercial Products, 2022-2035: Distribution by Application Area
21.6.4. Viral Vector and Plasmid DNA Manufacturing Market for Commercial Products, 2022-2035: Distribution by Key Geographical Regions
21.7. Current and Future Market Opportunity for Clinical Candidates
21.7.1. Viral Vector and Plasmid DNA Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Phase of Development
21.7.2. Viral Vector and Plasmid DNA Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Type of Vector Manufactured
21.7.2.1. AAV Vectors
21.7.2.1.1. AAV Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Phase of Development
21.7.2.1.2. AAV Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Application Area
21.7.2.1.3. AAV Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.7.2.2. Adenoviral Vectors
21.7.2.2.1. Adenoviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Phase of Development
21.7.2.2.2. Adenoviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Application Area
21.7.2.2.3. Adenoviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.7.2.3. Lentiviral Vectors
21.7.2.3.1. Lentiviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Phase of Development
21.7.2.3.2. Lentiviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Application Area
21.7.2.3.3. Lentiviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.7.2.4. Retroviral Vectors
21.7.2.4.1. Retroviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Phase of Development
21.7.2.4.2. Retroviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Application Area
21.7.2.4.3. Retroviral Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.7.2.5. Plasmid DNA
21.7.2.5.1. Plasmid DNA Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Phase of Development
21.7.2.5.2. Plasmid DNA Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Application Area
21.7.2.5.3. Plasmid DNA Vector Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.7.3. Viral Vector and Plasmid DNA Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Application Area
21.7.4. Viral Vector and Plasmid DNA Manufacturing Market for Clinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.8. Current and Future Market Opportunity from Preclinical Candidates
21.8.1. Viral Vector and Plasmid DNA Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Type of Vector Manufactured
21.8.1.1. AAV Vectors
21.8.1.1.1. AAV Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Type of Animal Model Used
21.8.1.1.2. AAV Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Application Area
21.8.1.1.3. AAV Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.8.1.2. Adenoviral Vectors
21.8.1.2.1. Adenoviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Type of Animal Model Used
21.8.1.2.2. Adenoviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Application Area
21.8.1.2.3. Adenoviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.8.1.3. Lentiviral Vectors
21.8.1.3.1. Lentiviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Type of Animal Model Used
21.8.1.3.2. Lentiviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Application Area
21.8.1.3.3. Lentiviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.8.1.4. Retroviral Vectors
21.8.1.4.1. Retroviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Type of Animal Model Used
21.8.1.4.2. Retroviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Application Area
21.8.1.4.3. Retroviral Vector Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.8.1.5. Plasmid DNA
21.8.1.5.1. Plasmid DNA Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Type of Animal Model Used
21.8.1.5.2. Plasmid DNA Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Application Area
21.8.1.5.3. Plasmid DNA Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.8.2. Viral Vector and Plasmid DNA Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Type of Animal Model Used
21.8.3. Viral Vector and Plasmid DNA Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Therapeutic Area
21.8.4. Viral Vector and Plasmid DNA Manufacturing Market for Preclinical Candidates, 2022-2035: Distribution by Application Area
21.8.5. Viral Vector and Plasmid DNA Manufacturing Market for preclinical Candidates, 2022-2035: Distribution by Key Geographical Regions
21.9 Current and Future Market opportunity from Vector-Based Vaccines
21.9.1. Viral Vector and Plasmid DNA Manufacturing Market for Vector Based Vaccines, 2022-2035: Distribution by Scale of Operation
21.9.2. Viral Vector and Plasmid DNA Manufacturing Market for Vector Based Vaccines, 2022-2035: Distribution by Type of Vector Manufactured
21.9.3. Viral Vector and Plasmid DNA Manufacturing Market for Vector Based Vaccines, 2022-2035: Distribution by Therapeutic Area
21.9.4. Viral Vector and Plasmid DNA Manufacturing Market for Vector Based Vaccines, 2022-2035: Distribution by Key Geographical Regions
- PORTER’S FIVE FORCES ANALYSIS
22.1 Chapter Overview
22.2. Methodology and Assumptions
22.2.1. Key Parameters
22.2.2. Threats of New Entrants
22.2.3. Bargaining Power of Suppliers
22.2.4. Threats of Substitute Products
22.2.5. Rivalry Among Existing Competitors
22.3. Concluding Remarks
- KEY DRIVERS AND CHALLENGES
23.1. Chapter Overview
23.2. Viral Vector and Plasmid DNA Manufacturing Market: Key Drivers and Challenges
23.2.1. AAV Vectors
23.2.2. Adenoviral Vectors
23.2.3. Lentiviral Vectors
23.2.4. Retroviral Vectors
23.2.5. Plasmid DNA
23.3. Concluding Remarks
- SURVEY ANALYSIS
24.1. Chapter Overview
24.2. Analysis by Seniority Level of Respondents
24.3. Analysis by Type of Manufacturer
24.4. Analysis by Scale of Operation
24.5. Analysis by Type of Vector Manufactured
- CONCLUDING REMARKS
- EXECUTIVE INSIGHTS
26.1. Chapter Overview
26.2. Batavia Biosciences
26.2.1. Company Snapshot
26.2.2. Interview Transcript: Menzo Havenga, Chief Executive Officer and President
26.3. CEVEC Pharmaceuticals
26.3.1. Company Snapshot
26.3.2. Interview Transcript: Nicole Faust, Chief Executive Officer & Chief Scientific Officer
26.4. Vigene Biosciences
26.4.1. Company Snapshot
26.4.2. Interview Transcript: Jeffrey Hung, Former Chief Commercial Officer
26.5. Delphi Genetics
26.5.1. Company Snapshot
26.5.2. Interview Transcript: Cedric Szpirer, Former Executive and Scientific Director
26.6. Clean Cells
26.6.1. Company Snapshot
26.6.2. Interview Transcript: Olivier Boisteau, Strategy Direction and Executive Board Member, Laurent Ciavatti, Former Business Development Manager and Xavier Leclerc, Head of Gene Therapy
26.7. Novasep
26.7.1. Company Snapshot
26.7.2. Interview Transcript: Alain Lamproye, Former President of Biopharma Business Unit
26.8. Amsterdam BioTherapeutics Unit (AmBTU)
26.8.1. Organization Snapshot
26.8.2. Interview Transcript: Joost van den Berg, Former Director
26.9. MGH Viral Vector Development Facility, Massachusetts General Hospital
26.9.1. Organization Snapshot
26.9.2. Interview Transcript: Bakhos Tannous, Director
26.10. Translational Vector Core, University of Nantes
26.10.1. Organization Snapshot
26.10.2. Interview Transcript: Eduard Ayuso, DVM, PhD, Scientific Director
26.11. CJ PARTNERS
26.11.1. Company Snapshot
26.11.2. Interview Transcript: Colin Lee Novick, Managing Director
26.12. ACGT
26.12.1. Company Snapshot
26.12.2. Interview Transcript: Semyon Rubinchik, Scientific Director
26.13. Richter-Helm
26.13.1. Company Snapshot
26.13.2. Interview Transcript: Astrid Brammer, Senior Manager Business Development
26.14. Plasmid Factory
26.14.1. Company Snapshot
26.14.2. Interview Transcript: Marco Schmeer, Project Manager and Tatjana Buchholz, Former Marketing Manager
26.15. Waisman Biomanufacturing
26.15.1. Company Snapshot
26.15.2. Interview Transcript: Brian M Dattilo, Business Development Manager
26.16. EFS-West Biotherapy
26.16.1. Company Snapshot
26.16.2. Interview Transcript: Beatrice Araud, ATMP Key Account Manager
26.17. GEG Tech
26.17.1. Company Snapshot
26.17.2. Interview Transcript: Nicolas Grandchamp, R&D Leader
26.18. Polypus Transfection
26.18.1. Company Snapshot
26.18.2. Interview Transcript: Géraldine Guérin-Peyrou, Director of Marketing and Technical Support
26.19. Vive Biotech
26.19.1. Company Snapshot
26.19.2. Interview Transcript: Naiara Tejados, Head of Marketing and Technology Development
26.20. Independent Consultant
26.20.1. Interview Transcript: Jeffery Hung
To view more details on this report, click on the link:
You may also be interested in the following titles:
You may also like to learn what our experts are sharing in Roots educational series:
|
Unlocking the Potential of Digital Twins in Healthcare: Benefits, Challenges and Opportunities |
About Roots Analysis
Roots Analysis is a global leader in the pharma / biotech market research. Having worked with over 750 clients worldwide, including Fortune 500 companies, start-ups, academia, venture capitalists and strategic investors for more than a decade, we offer a highly analytical / data-driven perspective to a network of over 450,000 senior industry stakeholders looking for credible market insights.
Contact:
Ben Johnson
+1 (415) 800 3415
+44 (122) 391 1091